Glifosatas
-
Aktualiausios mokslinės temos
- Užkirsti kelią vėžiui
- Odą jautrinančios cheminės medžiagos
- Per- ir polifluoralkilo medžiagos (PFAS)
- Mikroplastikai
- Aikštynų dangos granulės ir mulčias
- Tatuiruočių dažai ir ilgalaikis makiažas
- Glifosatas
- Endokrininę sistemą ardančios medžiagos
- Bisfenoliai
- Cheminių medžiagų strategija tvarumui užtikrinti
- Bandymai su gyvūnais pagal REACH
- Ftalatai
- Biocides
- Švinas
- Research to enhance protection of our health and environment
Glifosatas
Įdėtųjų programų
Glifosatas

Glifosatas – tai viena plačiausiai pesticiduose naudojamų veikliųjų medžiagų, neleidžiančių nepageidaujamiems augalams augti greta pasodintų kultūrų arba sunaikinančių tokius augalus arba jų dalis. Šios cheminės medžiagos dažnai vadinamos herbicidais arba piktžoles naikinančiomis medžiagomis.
Glifosatas naudojamas žemės ūkyje ir sodininkystėje siekiant prieš sėją išnaikinti piktžoles. Tais atvejais, kai auginami glifosatui atsparūs genetiškai modifikuoti augalai, cheminė medžiaga taip pat naudojama po sėjos naikinant tarp kultūrų augančias piktžoles. Tačiau tokia praktika Europos Sąjungoje netaikoma.
ECHA prižiūri, kaip įgyvendinamas su cheminių medžiagų ir mišinių klasifikavimu ir ženklinimu susijęs teisės aktas. Šiuo teisės aktu reglamentuojama cheminių medžiagų vertinimo ir ženklinimo tvarka, atsižvelgiant į pavojingas savybes, kurių gali turėti tokios medžiagos (pvz., nuodai, degios cheminės medžiagos, ėsdinančios cheminės medžiagos ir pan.).
Dėl tam tikrų cheminių medžiagų, pvz., pesticidų, klasifikavimo sprendimas gali būti priimamas ES lygmeniu, tuomet jis galioja visoje Europoje. Praktiškai tai reiškia, kad bus reikalaujama naudoti atitinkamą ženklinimą, siekiant įspėti vartotojus apie kiekvieną produktą, kuriame yra cheminė medžiaga.
In 2019, the Glyphosate Renewal Group (GRG) formally applied to renew the approval of glyphosate for use after the current approval expires at the end of 2022.
Four EU Member States – France, Hungary, the Netherlands and Sweden – formed the Assessment Group on Glyphosate (AGG). They assessed the application and submitted their reports to ECHA and the European Food Safety Authority (EFSA).
In parallel with EFSA's assessment, ECHA's Committee for Risk Assessment, (RAC), reviewed the classification of glyphosate. RAC is mandated to review only the hazardous properties of the substance, and not the likelihood of exposure to glyphosate.
RAC's assessment started in 2021. A consultation on the classification proposal was launched in September 2021. RAC's final opinion was published in July 2022 and it will feed into EFSA's risk assessment.
Based on EFSA's risk assessment, the European Commission will decide on the renewal of the approval of glyphosate.
Latest updates
28 November 2023
Commission's press release - Glyphosate: Commission adopts a renewal for 10 years
6 July 2023
EFSA's press release - Glyphosate: no critical areas of concern; data gaps identified
11 July 2022
Exchange of views between the European Parliament's Committee on the Environment, Public Health and Food Safety (ENVI), ECHA and the European Food Safety Authority (EFSA)
5 July 2022
Opinion of the Committee for Risk Assessment (RAC) published
30 May 2022
Glyphosate: no change proposed to hazard classification
ECHA's Committee for Risk Assessment (RAC) agrees to keep glyphosate’s current classification as causing serious eye damage and being toxic to aquatic life. Based on a wide-ranging review of scientific evidence, the committee again concludes that classifying glyphosate as a carcinogen is not justified.
10 May 2022
Glyphosate: EFSA and ECHA update timelines for assessments
The consultations carried out by EFSA and ECHA on the draft assessments of glyphosate attracted an unprecedented number of comments, confirming the high level of interest in this substance. Such a level of public participation underlines the importance of transparency in the evaluation of active substances in the EU.
The input received from the consultations, together with the replies received by EFSA from the applicant (the Group on the Renewal of Glyphosate) in response to its request for additional information, added a significant amount of information to a dossier that already contained far more scientific data than are usually available for such assessments.
This additional information will be thoroughly considered by the Assessment Group on Glyphosate (AGG), composed of four EU Member States – France, Hungary, the Netherlands and Sweden – that is now updating their initial draft renewal assessment report (dRAR).
Against this background, EFSA and ECHA have revised the timeline for the remaining steps in the re-evaluation process.
30 March 2022
Ad hoc consultation on glyphosate
New articles potentially relevant to the classification of glyphosate have been published. They relate to the following hazard classes:
- respiratory sensitisation,
- specific target organ toxicity - single exposure (respiratory irritation),
- germ cell mutagenicity,
- carcinogenicity,
- reproductive toxicity and
- hazardous to the aquatic environment.
ECHA is looking for comments related to these by 14 April.
2 December 2021
Glyphosate consultations: over 400 submissions collected
The parallel consultations on glyphosate held by EFSA and ECHA closed on 22 November 2021.
During the two-month consultation, all interested parties had access to the scientific evaluations prepared by the national competent authorities of Hungary, France, the Netherlands and Sweden, known collectively as the Assessment Group on Glyphosate (AGG).
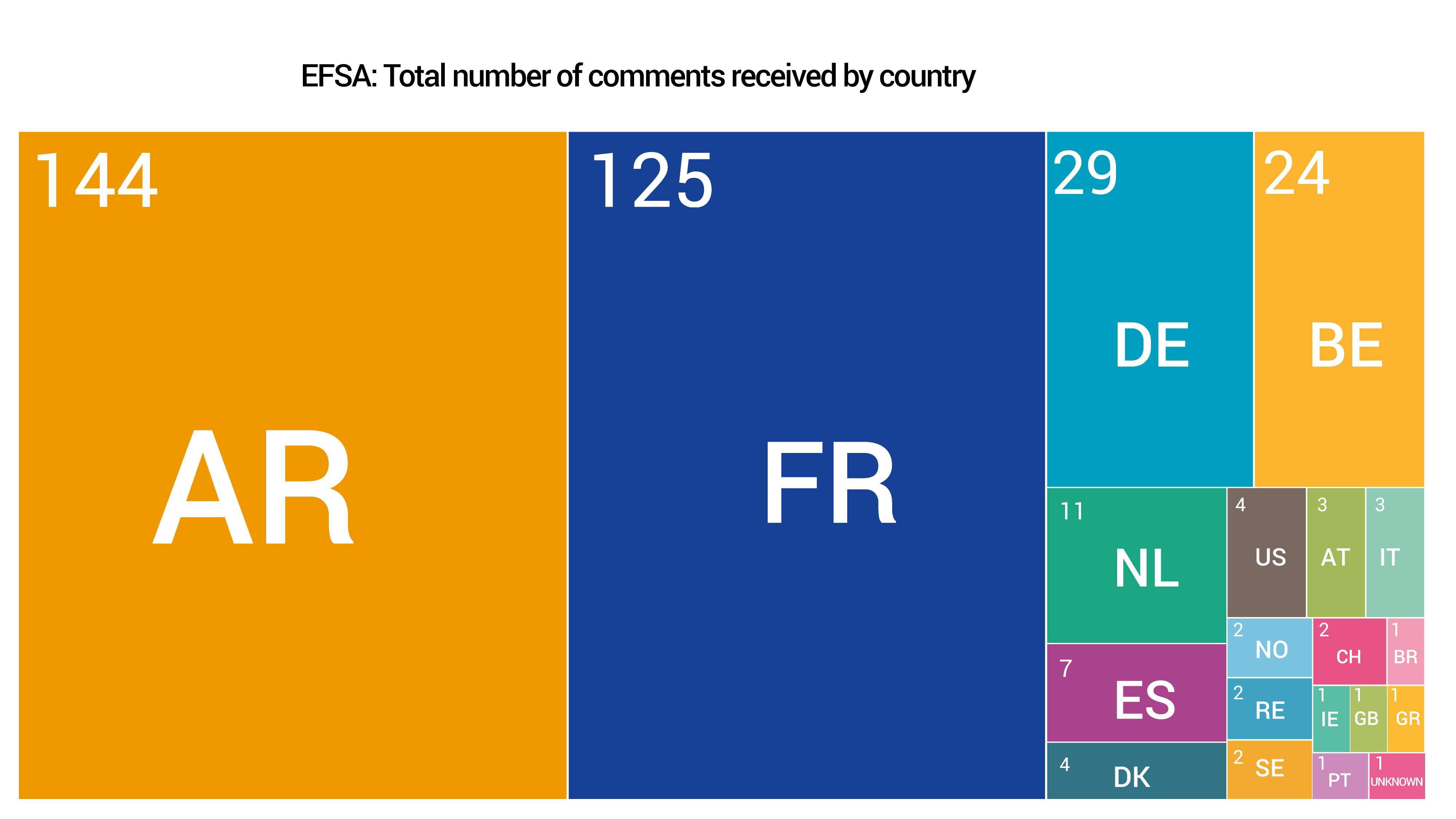
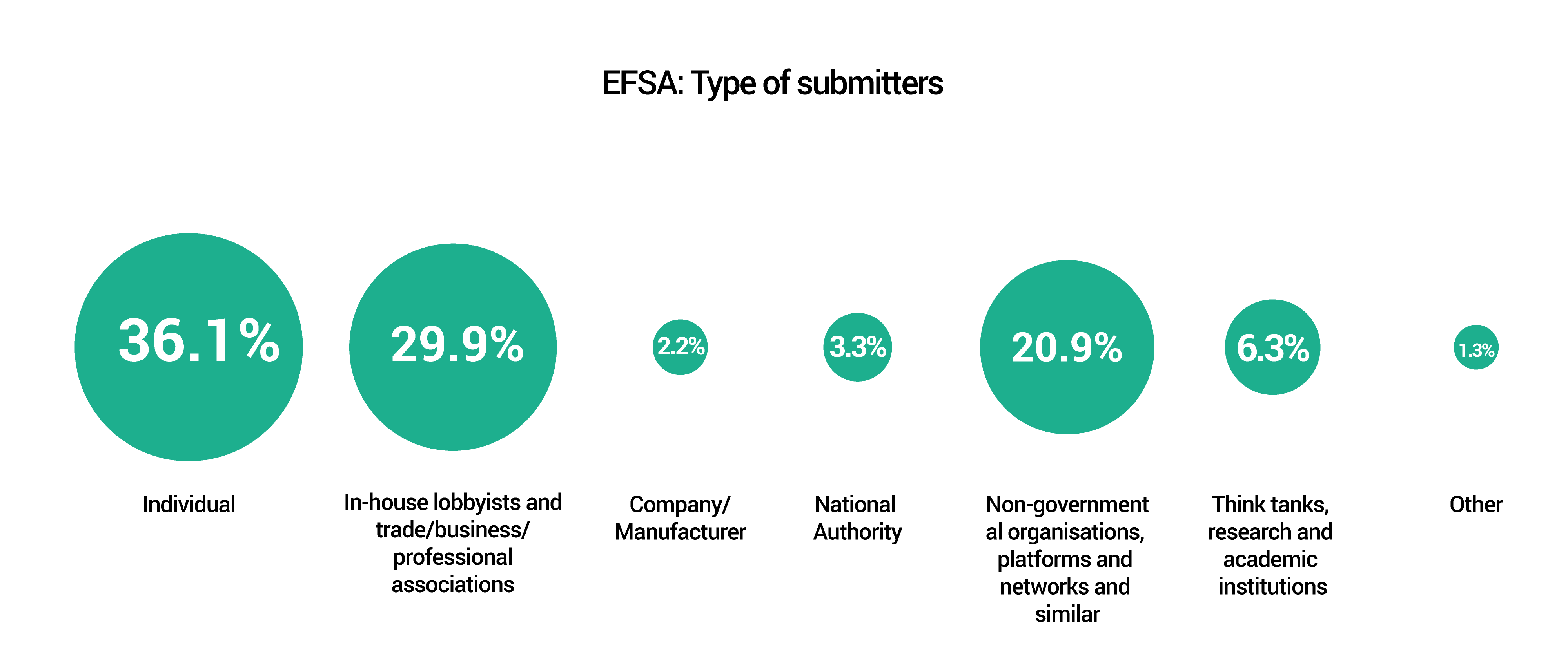
A total of 416 submissions were received through the two consultations from within and outside the EU, the largest number coming from Argentina.
These submissions – some carrying multiple comments – are publicly available and can be found on the EFSA and ECHA websites.
The graphs below provide a snapshot of the submissions received.


Next steps in the EU re-evaluation of glyphosate
The comments and observations received through the consultations have been shared with the AGG, who will consider and respond to them.
For the classification process, the AGG will provide its responses to ECHA’s Committee for Risk Assessment (RAC). The RAC will develop its opinion on the classification of glyphosate under the Classification, Labelling and Packaging (CLP) Regulation. This is currently expected in May/June 2022.
For EFSA’s assessment, following the AGG’s evaluation of the comments received through the consultation, a clock stop could be applied to request additional information from the Glyphosate Renewal Group (GRG). An updated assessment (RAR) will then be produced by the AGG and will be sent to EFSA to proceed with the peer review. EFSA will take the outcome of ECHA’s opinion into account in its peer review, which is expected to be finalised in the second half of 2022.
Regular updates on the work can be found on the EFSA and ECHA websites.
- Assessment Group on Glyphosate
- Glyphosate on EFSA website
- Glyphosate consultation by ECHA
- Glyphosate consultations by EFSA
23 September 2021
Glyphosate: ECHA and EFSA launch consultations
ECHA and the European Food Safety Authority (EFSA) have started parallel consultations on the initial scientific evaluations of glyphosate. The consultations will run for 60 days and all interested parties are encouraged to contribute.
30 August 2021
EFSA and ECHA will launch parallel consultations on the Renewal Assessment Report (RAR) and the Harmonised Classification and Labelling (CLH) report on glyphosate by 24 September 2021. All interested parties will be able to contribute to the ongoing scientific assessment by submitting relevant comments and information. The consultation will remain open for 60 days.
10 August 2021
The Assessment Group on Glyphosate submitted updated versions of the Renewal Assessment Report (RAR) and the Harmonised Classification and Labelling (CLH) report to EFSA and ECHA respectively. The two EU regulatory agencies will now carry out the administrative work that is needed before the documents are made available for consultation. The parallel consultations were announced for the first week of September 2021 but will be rescheduled. The new date will be communicated in due course.
Assessment Group on Glyphosate
15 June 2021
Glyphosate: EU regulators begin review of renewal assessments
ECHA and the European Food Safety Authority (EFSA) have received a draft assessment of glyphosate carried out by four EU Member States and will now begin to consider the findings. Glyphosate – the most widely used herbicide in the world – is currently authorised for use in the EU until December 2022.
FURTHER INFORMATION
- Glyphosate: no change proposed to hazard classification
- Harmonised classification and labelling report
- Glyphosate: questions and answers [PDF][EN]
ECHA's responses to stakeholder reactions on glyphosate assessment
- ECHA's and EFSA's input to Aarhus review
- Response from ECHA and EFSA to the European Commission related to publication from Générations Futures [PDF][EN]
- Response from ECHA and EFSA to the European Commission related to letter from NGOs [PDF][EN]
- Response from ECHA and EFSA to Members of the European Parliament [PDF][EN]
- Response to the report from Health and Environment Alliance (HEAL) [PDF][EN]
- Response to a specific question from ENVI Committee exchange of views [PDF][EN]
European Commission
EFSA
Stakeholder presentations to RAC meeting, March 2022
- Assessment Group on Glyphosate AGG [PDF][EN]
- Health and Environment Alliance HEAL [PDF][EN]
- European Environmental Bureau EEB [PDF][EN]
- ClientEarth [PDF][EN]
- Bayer [PDF][EN]
- Glyphosate Renewal Group [PDF][EN]
- EFSA [PDF][EN]
ECHA's process in 2017
- Glyphosate not classified as a carcinogen by ECHA, 15 March 2017
- ECHA's role in assessing glyphosate, 7 July 2016


