Dossier submission for active substance approvals
Dossier submission for active substance approvals
Companies will use electronic submission tools for applying for an approval for a new active substance or a renewal of their approval. Applications are submitted through R4BP 3 as a IUCLID file.
This graph shows an overview of the dossier submission process.
Steps
Dossier submission proceeds in several steps. Each step needs to be completed before the application can move to the next step. It is important that the applicant makes sure that all applicable deadlines are met, otherwise the application will be rejected during the course of the process.
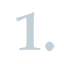
ECHA checks that the application and data have been submitted in the correct format.
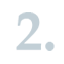
The applicant pays the related fees to ECHA within 30 days from the invoice date.

ECHA accepts the application and the evaluating competent authority has 30 days to validate the application (completeness check).

The applicant pays the related fees to the evaluating competent authority within 30 days.

If the dossier is deemed to be incomplete, the evaluating competent authority will ask for the missing information and the applicant will have 90 days to provide it.

Evaluation of the dossier starts.
Actors
The main actors in the dossier submission process are:
Applicants
Applicants have the responsibility to provide dossiers with all relevant information on their active substances and to provide additional information if requested by the evaluating competent authority. Applicants are responsible for the quality of the data in their dossiers.
ECHA
ECHA is responsible for ensuring that the information in the dossiers is in a correct format. ECHA also makes sure that the submission process proceeds within the set timelines.
Evaluating competent authority
The evaluating competent authority is responsible for carrying out the validation of the application dossiers and subsequently for the evaluation of the dossiers submitted by the applicants. Applicants can choose which Member State they would like to have evaluating their dossiers. This is finally decided in agreement with the Member State.



